 
The protons in the nucleus, on the other hand, have the same charge on them and tend to repel each other. Other and therefore electrons get pulled toward the protons. Under normal circumstances, the electrons cannot escape from the atom since positive and negative electrical charges attract each Particles in an atom or molecule together. You may now ask what keeps these different The protons and the neutrons in the center make up the nucleus of the atom. (grey) which circle around the protons and neutrons. It consists of two protons (red), two neutrons (blue) and two electrons one of the atoms contained in helium balloons. The Animation on the right shows a helium atom, i.e. One is that electrons carry one negative electrical charge, while protons carry one positive electrical charge. Electrons, protons and neutrons have different physical properties. Of neutrons, however may be different from the number of electrons or protons. The number of electrons in an atom always equals the number of protons in an atom. The atoms and molecules in a gas are made up of electrons, protons and neutrons. steam rising out of a pot of boiling water or a hot spring (see picture below).Small to see with the naked eye, and is made up of just a few atoms. Tubes or the brilliant Aurora Borealis (or Northern Lights) sometimes visible in the night sky near the north or south Pole.Ī gas is made up of a lot of tiny, little particles called molecules. It can glow and shimmer, as for example the bright, pink haze in neon Therefore they can conduct as their ions are free to move.A plasma is a gas that contains electrically charged gas particles. Ionic compounds dissolve in water easily, when they do this their lattice breaks up completely. This is a regular arrangement of metal and non-metal ions which creates compounds with very high melting points which conduct when molten or in solution but NEVER when solid. Ionic compounds form what is known as a lattice structure. When these two charged particles come together they form an ionic bond because the positive magnesium ion is attracted to the negatively charged chlorine ion. Non-metals form negative ions because they gain electrons to become stable. They change into ions with a two positive charge. To become stable it must lose its two outer electrons to obtain a full outer energy level.Ītoms are neutral because they have equal numbers of protons and electrons however, when they lose two electrons they are no longer neutral. The ionic bond is the force of attraction between the oppositely charged ions - a positively charged metal ion and a negatively charged non-metal ion.įor example Magnesium (Mg) has the electron arrangement 2,8,2. Sodium will lose an electron and form a positive ion.Ĭhlorine will gain an electron and form a negative ion. 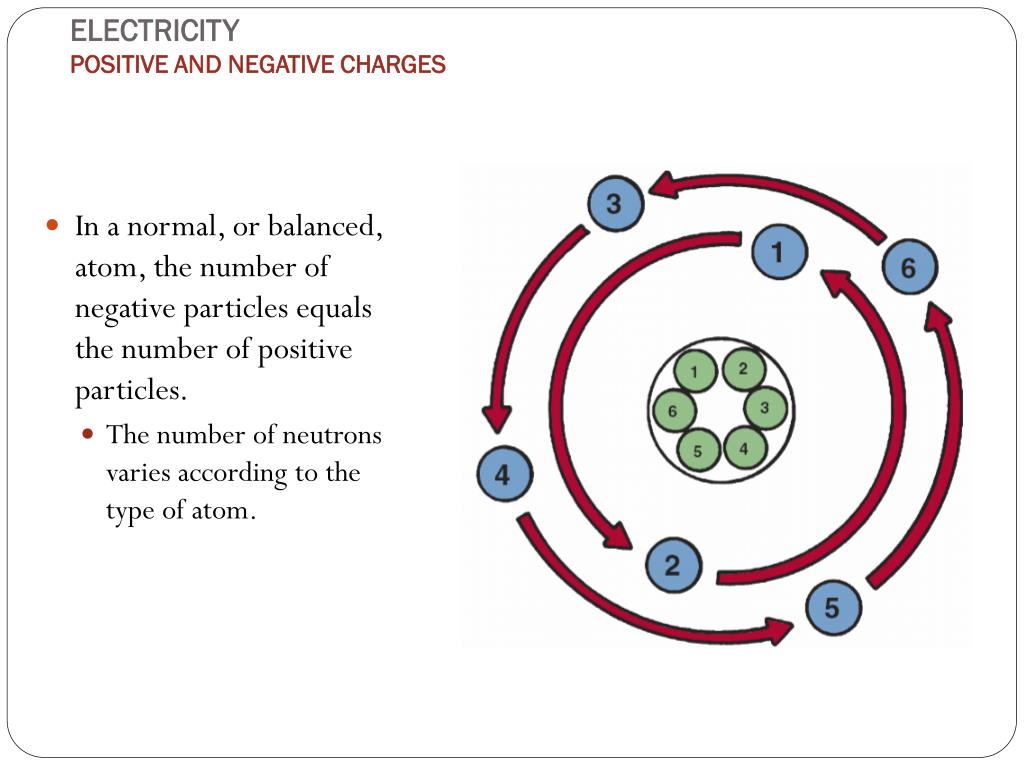
Outer electrons are transferred from the metal to the non-metal. Ionic bonds are formed between a metal and non-metal, for example, sodium chloride. Substances that consist of covalent molecules are usually gases or liquids at room temperature. Covalent compounds do not conduct electricity. A molecule is a group of atoms held together by covalent bonds. 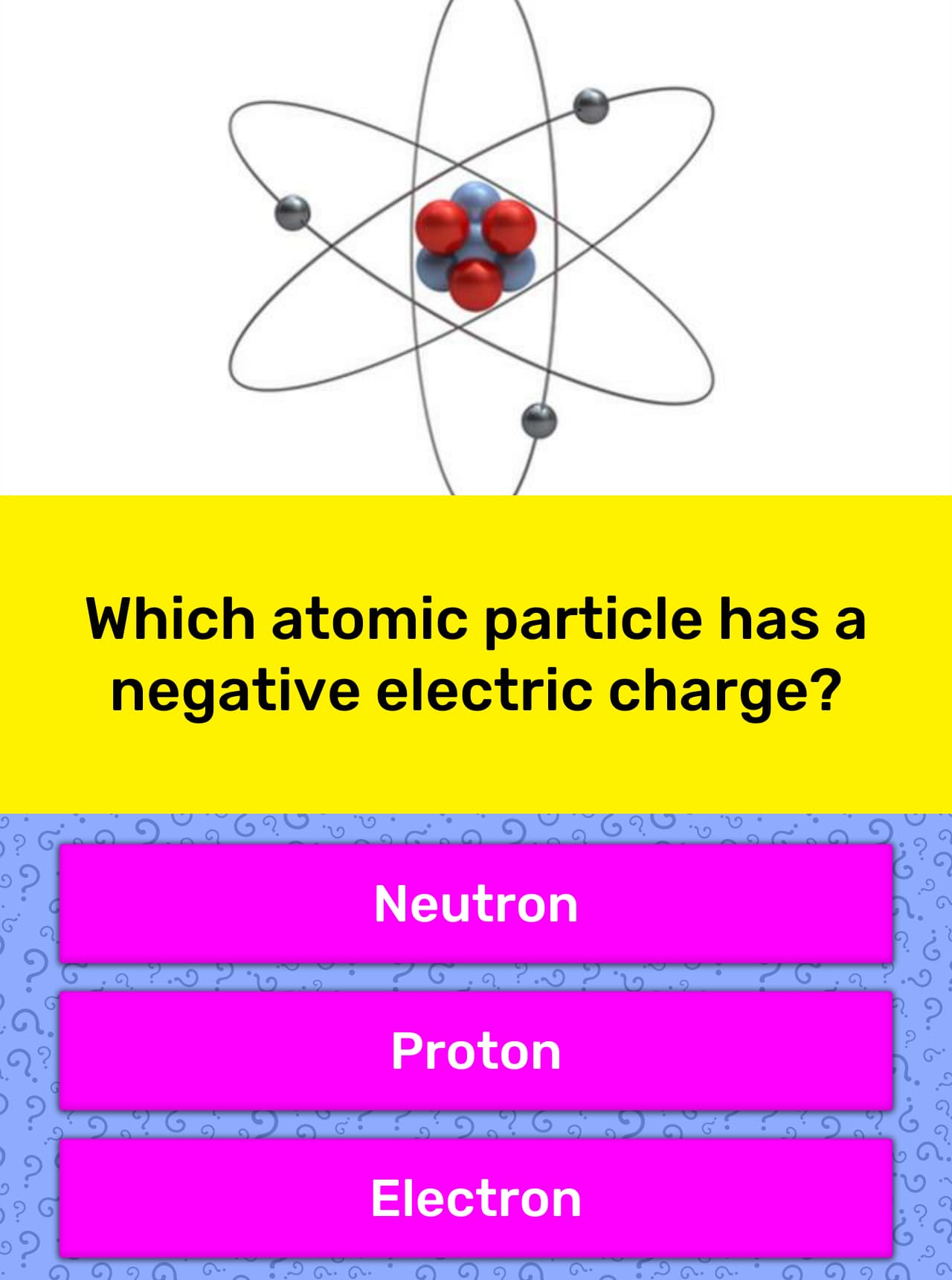
Two atoms sharing a pair of electrons.Ī covalent bond happens when the positive nuclei from two different atoms are held together by their common attraction for the shared pair of electrons held between them.Ītoms that share pairs of electrons form molecules. A covalent bond is a shared pair of electrons between two non-metal atoms, for example carbon dioxide. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
